The shortage of respiratory ventilators in the face of the Covid-19 pandemic has led to numerous efforts aimed at turning hand-operated plastic pouches called bag-valve resuscitators into makeshift ventilators.
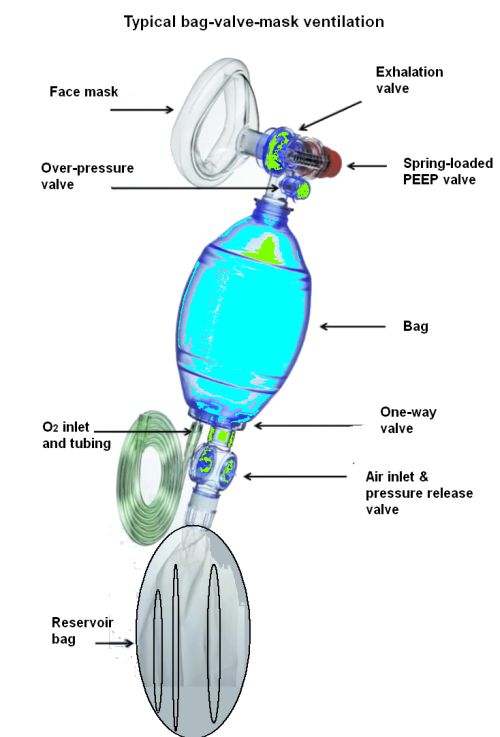
The resuscitators, also called Ambu bags, are generally on-hand in large quantities. They are designed to be operated by hand, by trained technicians, to inflate patient lungs until a better device such as a ventilator becomes available. In operation, the technician inserts a tube into the patient’s airway and then pumps air into the lungs by squeezing and releasing the flexible pouch.
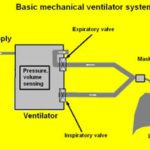
The usual approach to turning bag resuscitators into make-shift ventilators is to add a mechanical means of squeezing the bag. Teams at facilities that include M.I.T., UC San Diego, and the University of Minnesota all have efforts focused around this basic idea.

Problems can arise because a manual resuscitator force-inflates the lungs. Possible complications include air inflating the stomach, lung injuries from over-stretching, and lung injuries from over pressurization. In particular, gastric inflation can lead to vomiting with some stomach contents ending up in the lungs. Over-stretched lungs can lead to adult respiratory distress syndrome that requires prolonged mechanical ventilator support in an ICU and, according to Wikipedia, is associated with poor survival. Other problems include hyperventilation, either too many breaths per minute or breaths that are too large.
One might suspect that many such difficulties arise from the mistakes made by technicians manually squeezing the bag. If so, the regular repeatable action of a mechanical squeezer could be an improvement. However, it is clear from some of the carefully worded statements made by teams working on these devices that potential hazards are top-of-mind. A key component in any mechanized resuscitator will be a pressure sensor that monitors lung inflation. One might surmise that there are likely to be discussions among the developers of these devices about whether pressure monitoring alone is enough to head off the well-known potential complications.
Of course, none of these efforts would be acceptable for use in medical facilities were it not for recent emergency use authorizations issued by the Federal Drug Administration. An important point to note about these authorizations is that they allow modifications that turn existing devices into respiratory ventilators, but only if the equipment being modified has already gone through FDA approval. The Ambu bags used as a basis for the projects at MIT and other institutions have all gotten FDA approval, making them fair game for modifications.
The same can’t be said for most of the open-source projects with the same goals.

Are these devices more dangerous than the virus, or is the FDA more dangerous than the virus?
The objections raised are simple to overcome.
1. Cyclic timers on the squeezers
2. Aoppripriate pressure relief valves in the supply stream
3. Pressure sensors to monitor the air pressure in the supply stream
4. Set timing controls for the supply stream and the return
5. Alarms for: pressures, flow rates, stoppages, other critical parameters
6. Data-logger
7. Display
8. Control buttons
In addition lung inflation can be monitored
It is interesting to note that almost as a post-script, the Author has mentioned that all devices based on the Ambu Bag, including the MIT product are all FDA approved, plus these develiiopments too place a while ago, I might add.